Bioflavonoids - pycnogenol, proanthocyanidin and quercetin
Substances with the above two names are widely marketed as anti-oxidant vitamin pills without revealing the simple fact that both names refer essentially to tannins which are ubiquitous in plants!
(See molecular structures illustrated below!) Here are some homepages dealing with this:
Pycnogenols 1: or Pycnogenols 2: or Pycnogenols 3:
Bioflavonoids have become of interest as prominent natural anti-oxidant free-radical scavengers, which also have a synergetic helper function with other anti-oxidants such as d-alpha tocopherol (Vitamin E), ascorbate (Vitamin C), carotene (Vitamin A) and Selenium in reducing the reputed 2000 mutational hits per cell per day caused by oxidation.
I currently take the following daily: d-alpha tocopherol 167 mg, calcium ascorbate 250 mg, selenium 100 mcg, carotene 1/2 cup carrot, and 30 mg proanthocyanidin. I find vitamin E very beneficial. Flavonoids however appear to be the principal natural antioxidant in the Dutch diet (see below). I currently supplement natural sources in fruit and nuts with proanthocyanidin, particularly if my fruit intake is intermittent. This provides for more vitamin E and a little less proanthocyanidin than current combined formulas. Subjectively proanthocyanidin does appear to act synergetically with vitamin E.
In addition I regularly take Japanese green tea, and include a red onion at intervals in the cooking, to provide a pulsed source of quercetin, along with other anti-carcinogens such as broccoli.
Pycnogenols: the term put forward by the Masquelier J, et. al. to designate flavan-3-ol derivatives, so as to distinguish these substances, upon chemical and pharmacological grounds, from the heterogenous group of flavonoid compounds.
Masquelier J, et al. Flavonoids and pycnogenols. Int J Vitam Nutr Res (1979) 49:307-311.
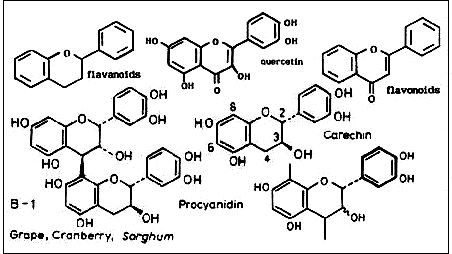
Proanthocyanidin: Fruedenberg and Weinges collectively designated all the colourless substances isolated from plants which when treated with acid form coloured anthocyanidins as proanthocyanadins. These include monomeric leucoanthocyanadins - flavan 3,4-diols such as catechin. Condensed proanthocyanadins are reserved for the various flavan 3,4-diol dimers and higher oligomers, including the procyanidins illustrated below. Fruit-bearing plants have proved to be particularly rich sources of oligomeric procyanidins. Isomeric variants of the dimers occur in the fruit, fruit pods, seeds and seed shells of a diverse range of plants including grape, cranberry, sorghum, apple, cocoa, quince, cherry, horse chestnut raspberry, blackberry, willow and poplar catkins, hops and rosehips just to mention a few. They are not confined to pine bark and grape seeds!
Harborne notes that in any plant tissue where proanthocyanidin synthesis occurs there is invariably found a range of proanthocyanidins - from the monomeric flavan-3-ols (catechins and gallocatechins) to the polymers. For each tissue the balance between these molecular forms is probably determined by the corresponding balance between the metabolic flux to the flav-3-en-3-ol and the rate of supply of the biological reductant NADPH. Tissues in which the flux is low and the NADPH supply is high will contain a range of proanthocyanidins of varying molecular size but those tissues in which the flux is high and the supply of NADPH is low will conversely contain predominantly the higher oligomeric forms -for example, seed coats. This facet of proanthocyanidin metabolism was noted in 1935 by the Robinsons who recognized the presence in plants of oligomers of quite different solubilities, the largest soluble oligomers having up to 20 units. The flavan C-C oligomeric bonds are acid labile. Heartwood and bark also provide rich sources of unusual proanthocyanidins.
Bate-Smith and Swain concluded in 1953 that these substance were equivalent in distribution and properties to that class of substance rather indefinitely defined in the botanical literature as "tannins" and were responsible for their properties such as precipitating proteins and polysaccarides and their astringent taste!
Bate-Smith E. and Swain T (1953) Chem. Ind. 377.
Fruedenberg K. and Weinges K. (1960) Tetrahedron 8 336.
The Flavonoids : advances in research, (1982) edited by J.B. Harborne and T.J. Mabry. London ; New York : Chapman and Hall.
Although the oligomeric forms are an order of magnitude more potent anti-oxidants, possibly because they better chellate Fe ions, tea possesses similar anti-oxidant properties. The 1994 and 1995 research from the International Camelia Society's medline articles illustrate a variety of properties of the tea bioflavonoids. The catechins include active (-)-epicatechin gallate and (-)-epigallocatechin gallate, which inhibit tumors and block collagenases.
Quercetanus (1607) was one of the first mediaeval scientists to recognise the existence of the essential oil industry for lavender and juniper oils leading to the discovery of another group - the terpenes. Quercetin has earned a controversial reputation, first as a mutagen and then as one of the strongest anti-cancer agents known, according to Terence Leighton, Ph.D., professor of biochemistry at the University of California at Berkeley.
Contrasting this is the literature indicating quercetin is a mutagen, which has given cause for concern when the related rutin is consumed in vitamin tablets and broken down to quercetin in the gut. Among the articles listed in the links below from the Longlevity Institute, you will find that the minimum mutagenic concentration of quercetin is 4 nmols, while experimental anti-cancer doses veer upwards from 4 micro mols. The average Dutch intake is around 16 mg/day (see below). So a rough estimate is 16 / 1000 g / 54.5 litres / 302 gm mol wt = 0.9 micro mols.
Quercetin is also associated with red wine . See also.
Red wine also contains polyphenol
antioxidants good for the heart
and another powerful anticancer substance
called resveratrol, a phytoalexin
It occurs very predominantly in red onions as well as broccoli and other vegetables. However if you think you should immediately take it off your menu because of its mutagenicity, bear in mind that in a study published by the National Cancer Institute and the Beijing Institute for Cancer Research in the People's Republic of China, persons with diets high in onions and garlic, which at dry weight have qercetin levels of 3% to 4%, have a 20-fold lower cancer risk than persons who don't eat these vegetables.
Note also that particular vegetables such as broccoli are renowned for other anti-cancer substances. Broccoli in particular has both mustard oils as allyl isothiocyanates and indoles such as carbinol which have their own toxicity, balanced by a pronounced anti-cancer potential.
Quercetin's anti-tumor action appears diverse and includes inhibition of inoculated cancer cells, chemical and virally induced cancers leukaemia and ovarian cancer. Related flavonoids also give protection against UV-B damage . Despite being a mutagen under test conditions, quercetin does not appear to be a carcinogen. in hamsters, or in rats although one should also explore Ames's data.
Note again the tea research:, which shows tea itself, despite having minor genotoxicity has significant inhibition of cancer and genetic damage associated with smoking.
Again, you will find in the thanksgiving dinner menu , tea is noted for its quercetin content.
Below are shown the molecular structures of flavanoid and flavonoid skeletons, quercetin a key flavonoid, and catechin and associated procyanadin dimers and polymer.